Fight gastric cancer by targeting Claudin 18.2
Fight gastric cancer by targeting Claudin 18.2

Gastric cancer (GC) is one of the most common and the 3rd deadly cancers worldwide, with a high frequency of recurrence and metastasis, and a poor prognosis. Effective treatment of GC remains challenges. Claudin 18.2 is considered as a new therapeutic target for advanced gastric cancer.
The normal and aberrant expressions of Claudin 18.2
arigo offers an excellent Claudin 18.2 specific antibody which is the best solution for precise and efficient detection of Claudin 18.2 expression.
|
Claudin 18.2 antibody (ARG67014) Claudin 18.1 predominantly expresses in lung whereas Claudin 18.2 displays stomach specificity. |
Claudin 18.2, an ideal anti-cancer target

Claudin 18.2 targeting therapy
Targeting Claudin 18.2 is a promising neo-approach for GC treatment as Claudin 18.2 is expressed in 50-80% of GC. Multiple therapeutic agents targeting Claudin 18.2, including mAbs, bispecific antibodies, chimeric antigen receptor T (CAR-T) cells, and antibody-drug conjugates (ADCs), have been developed. At present, a monoclonal antibody and chimeric antigen receptor engineered T (CAR-T) cells targeting Claudin 18.2 show promising results in clinical trials.
▶ Monoclonal antibodies targeting Claudin18.2
The clinical trials of several Claudin 18.2-targeted antibodies are ongoing. Zolbetuximab (IMAB362, claudixmab), the first developmental antibody-drug against Claudin 18.2, had been validated its ability to eliminate cancer cells in preclinical studies. Currently, Zolbetuximab is in phase III clinical trials for treating advanced/metastatic GC and advanced gastroesophageal junction (GEJ) cancer.
arigo offers the most sensitive M13 antibodies which are the best solutions to facilitate the screening of therapeutic antibodies by M13 phage display platform.

▶ CAR-T cells targeting Claudin 18.2
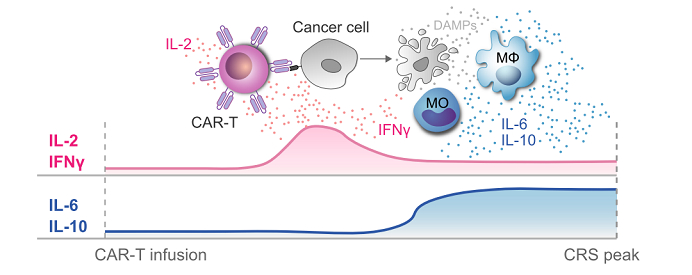
CAR-T-cell therapy is based on engineering of T lymphocytes to express chimeric antigen receptors (CARs), which enables the modified T lymphocytes to recognize and eliminate cancer cells. Early studies have explored the potential of Claudin 18.2-specific CAR T-cell therapy in GC and other cancers. The clinical efficacy and toxicity of multiple CAR-T-cell candidates are assessed by several phase I/II clinical trials.
As with CAR T-cell therapy in general, cytokine release syndrome (CRS) is a potential adverse effect that may make clinical application challenging. arigo offers the CRS Cytokine Multiplex ELISA Kit (ARG82969) which is the best solution to monitor the CAR-T induced CRS.
|
Human CAR-T / CRS Cytokine Multiplex ELISA Kit (ARG82969) The kit allows a concurrent measurement of 4 key cytokines |